Recall of Losartan
By Bryan Tan Van Hong
Publicity Associate at YPC. Currently working in Esyms, an online pharmacy platform and Registered Pharmacist at a retail pharmacy. Oh, and not to forget, pun enthusiast.
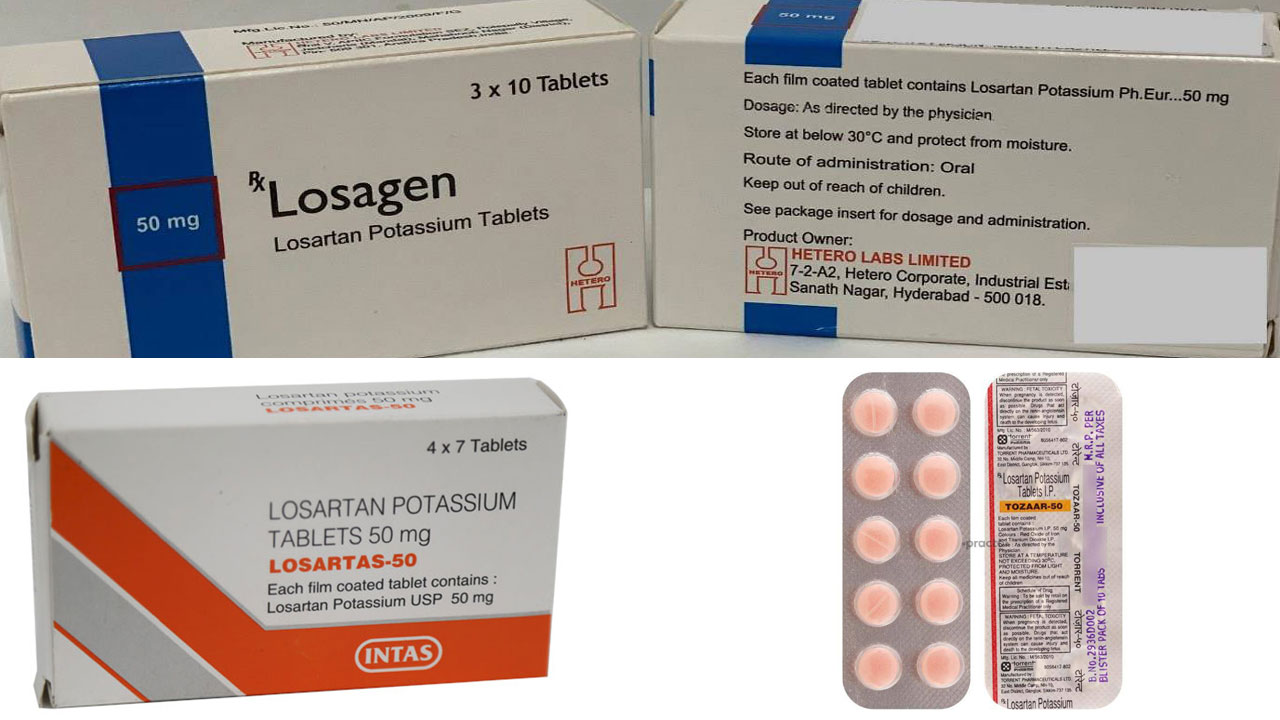
On early March, the Ministry of Health Malaysia released a statement regarding the recall of 7 types of medication containing the active ingredient Losartan. This came about after some of these angiotension II receptor blocker (ARB) were found to contain unacceptable amount of nitrosamine, a substance which is possibly carcinogenic, or has the potential to cause cancer.
The seven types of medications recalled were Losagen 50, Losagen 100, Losartas 50mg Tablets, Losartas 100mg Tablets, Tozaar Plus LS 50/12.5mg Tablet, Tozaar 50mg Tablet and Tozaar Tablet 100mg.
Deputy Minister Dr Lee Boon Chye urged those who are in possession of the aforementioned medication to return it to the supplier in exchange for other approved medication. Stern actions will also be taken on suppliers who continue to sell these medications.
However, there is no need to panic, says Dr Lee. “Do not panic if you have consumed the medications, as there are no short-term effects.”
This is not the first time losartan has been recalled from the market. Last year, a similar incident occured where the company which supplied Lostad HCT 50/12.5 mg was asked to cease its operation.
Be sure to visit your local pharmacy or clinic for any enquiries. Do not stop taking any medications on your own accord.
Related
2018 Management of Hypertension (5th Edition)
Last updated onWorld Pharmacists Day 2018 (20-21 October 2018)
Last updated onEvent: World Pharmacists Day 2018 Date: 20-21 October 2018 (Saturday and Sunday) Time: 10am – 10pm daily Venue: MyTown, Cheras, Kuala Lumpur Theme: Pharmacists, Your Medicines Expert Organised by Pharmaceutical Services Program Ministry of Health, ATM Ministry of Defense, MPS, MCPG, PhAMA, MOPI
NDMA Found in Ranitidine
Last updated onWritten by Bryan Tan – Marketing and Communications Associate at YPC, currently a PRP at a retail pharmacy, and pun enthusiast Recently, there has been news regarding the discovery of traces of N-Nitrosodimethylamin (NDMA) found in ranitidine medications. This was reported by the American-based Food and Drug Administration (FDA) as well as the Singaporean…
Community Pharmacy Logo
Last updated onThis logo was decided recently from a logo competition carried out by the Pharmaceutical Services Division of the Ministry of Health, in collaboration with MPS. Through a circular from the Pharmacy Enforcement Division of the Ministry of Health dated 30th January it is compulsory to display this logo at all community pharmacy premise. For existing…
Getting to know: First Malaysia-produced PharmD Graduate
Last updated onWe had the honour of meeting the first Malaysian-produced Doctor in Pharmacy (PharmD) graduate. Dr. Chong Chin Pin, or more commonly known as CP by those who know him personally, who is also YPC’s treasurer, recently graduated and received the PharmD title. We sat down and spoke to him in hopes of obtaining a clearer…
Malaysian Clinical Pharmacy Symposium (MCPS) 2.5
Last updated onWritten by Tang Jia Wen, commitee member of MCPS 2.5 On 27 July 2019, the Malaysian Clinical Pharmacy Symposium (MCPS) 2.5 – A Cancer Therapy Counselling Workshop was held in International Medical University (IMU). It had successfully gathered pharmacists from different backgrounds and provided a conducive platform for them to exchange ideas and thoughts…
Others
COVID-19 Vaccine Updates: Latest Worldwide Developments, Malaysia’s Involvement, and Barriers to Vaccine Success
Last updated onWHO to look into drug pricing?
Last updated onOur health ministry has urged the World Health Organisation (WHO) to look into drug pricing by major drug companies. This came about amidst the pressure on the ministry to utilise Compulsory Licensing (CL)* to gain access to generic versions of a Hepatitis C medication. “I hope the international community could look at how pharmaceutical companies…
Vaccine, or Vacc-out?
Last updated onAn infant aged 2 years and 1 month recently succumbed to a very preventable disease – diptheria. It was reported that the infant was never once immunised since birth and passed away on 21 February due to severe diptheria with multiorgan failure. Diptheria is caused by the Corynebacterium diphtheriae bacteria and can be communicated via…
Compulsory Vaccination a No-Go?
Last updated onAmidst the governement’s plans to look into compulsory vaccination, The United Nations’ children’s agency has said that this is not the best way to address life-threatening diseases such as measles and diphteria. Marianne Clark-Hattingh, Unicef’s representative in Malaysia said that mandatory laws do not always increase vaccination coverage, according to evidence. After the recent death…
Poison Found in Cosmetic Product
Last updated onWritten by Bryan Tan – Marketing and Communications Associate at YPC, currently a PRP at a retail pharmacy, and pun enthusiast This is not an april fool’s joke! The Desk of the Director General of Health released a statement on 1 April on 2 cosmetic products found to contain a scheduled poison. The 2…
Coronavirus Declared as Global Emergency by WHO
Last updated onThe recent coronavirus outbreak took us by surprise. This storm came as sudden as a midnight deluge, and it seems like it is here to stay. Admittedly, even I myself am wearing a mask as I type this. What seem to otherwise be commonplace items such as face masks and hand sanitizers are now scarce…